New research has bolstered the argument that a century-old pain management technique is effective for treating post-traumatic stress disorder.
Results of a clinical trial published online Wednesday by JAMA Psychiatry show that stellate ganglion block, or SGB, injections reduced PTSD symptoms at roughly twice the rate of a placebo, resulting in a noticeable improvement for those who received them.
The study of 108 active duty military personnel is the largest to date to test the treatment, which involves injecting a mild anesthetic — in this case, ropivicaine — into a bundle of nerves in the neck that serve the neck, head, arms and upper chest.
Two injections were given to participants two weeks apart, with two-thirds of the patients receiving SGB shots and one third receiving a placebo. All patients took the Clinician-Administered PTSD Scale assessment, called the CAPS-5, before their first injection and then after eight weeks.
Those who receive the SGB improved on average 12.6 points on the CAPS-5, while those who received the sham injection improved by 6.1 points. A 10-point improvement is “enough that the person should notice a significant improvement in their life,” according to Kristine Rae Olmsted, a research epidemiologist with RTI International, the nonprofit research institute that conducted the Army-funded study.
“The literature [on SGB] up until now has been pretty positive and the anecdotes … were pretty strongly positive. But we were all surprised by the magnitude of the difference,” Rae Olmsted said.
The research reinforces previous case studies of the effectiveness of SGB for treating PTSD. A review published in the journal Military Medicine in 2014 showed that nearly 80 percent of patients receiving the treatment reported a reduction in their PTSD symptoms like anxiety, hyperarousal, sleep disturbances and depression after one shot.
The therapy has been used for years to treat chronic pain, uncontrolled sweating, hot flashes and pain caused by shingles and angina. A Chicago-area pain management specialist, Dr. Eugene Lipov, began testing it to treat PTSD in 2008. Navy and Army physicians also began trying the procedure, with Army doctors using it at Walter Reed Army Medical Center in Washington and Navy physicians launching a research study at Naval Medical Center San Diego in 2012.
But that study, involving 42 service members, showed that SGB performed no better than a sham treatment, leading is primary investigator, Dr. Robert McLay, NMCSD’s mental health research director, to conclude that previous improvements reported in case studies were likely attributable to the placebo effect and more research was needed on the effectiveness of more than one shot.
In a rebuttal, Lipov, who remains a strong proponent of the use of SGB to treat PTSD, noted that McLay’s study members were involved in medical boards that were determining their disability ratings and thus had a financial incentive not to report improvement.
This study appears to debunk that conclusion.
“Now we have a strong study with the highest level of evidence that shows that SGB can really help PTSD symptoms,” said retired Army Col. Sean Mulvaney, who led the study with Rae Olmsted and first began exploring SGB as a potential treatment for military troops with PTSD in 2010. “I hope this study finally helps patients get the relief they need.”
RELATED
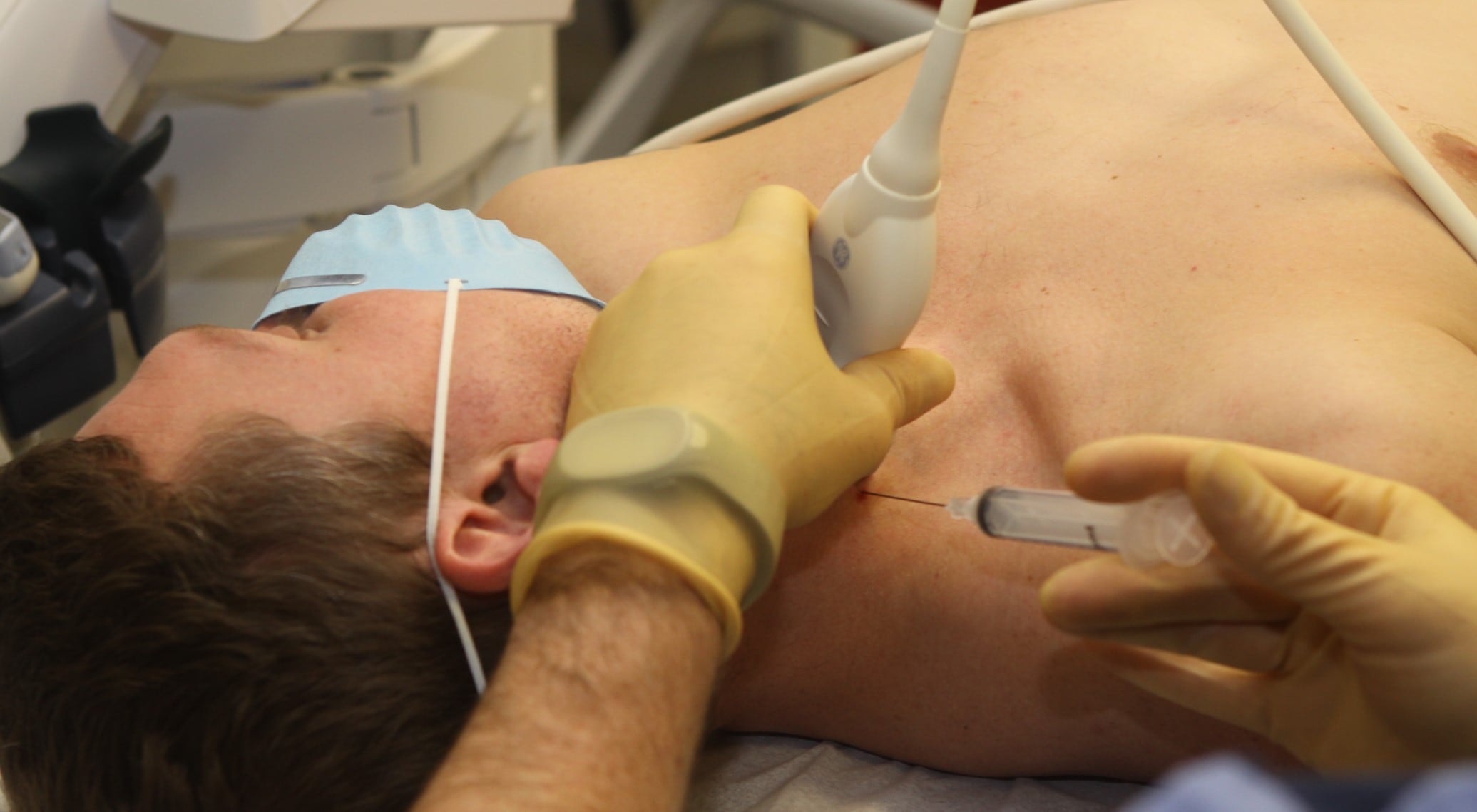
The mechanism for how SGB injections work to address PTSD symptoms isn’t clear-cut. Researchers say that anesthetizing the group of sympathetic nerves influences the “fight or flight” impulse and interrupts pain signals.
“Anesthetizing the ganglion blocks nerve impulses temporarily. We still don’t know how SGB works to improve PTSD symptoms, but now we know that it does,” Rae Olmsted said.
The procedure carries very little risk, according to Rae Olmsted. A trained physician conducts the procedure in a medical facility, using ultrasound to locate the stellate ganglion and inject the medication. Side effects include temporary bruising, drooping eye, tearing and hoarseness.
The study took place at Womack Army Medical Center, Fort Bragg, North Carolina, Tripler Army Medical Center, Honolulu, Hawaii, and Landstuhl Regional Medical Center, Germany. RTI International is planning a more extensive study, also funded by the U.S. Army, to take place at five military facilities that will follow participants for a year after the treatment to explore long-term effectiveness and side effects, Rae Olmsted said.
She added that the Defense Department is being wise in thoroughly researching the treatment before offering it more broadly. “We subject service members to enough without potentially subjecting them to treatments we aren’t sure work.”
She added, however, that patients can speak to their doctors if they are interested in receiving the treatment. More than 2,000 service members and civilians have received SGB injections to treat PTSD.
Patricia Kime is a senior writer covering military and veterans health care, medicine and personnel issues.





